General properties and Specificities
1. Basic concepts
Since its introduction, approximately 15 years ago, the Implantable Loop Recorder (ILR) has become the reference tool for the diagnosis of unexplained syncope. Syncope is a major public health concern as, in a lifetime, between 40 and 50% of the general population suffer ≥1 episode(s), which account for 1 to 6% of all hospitalisations. While a thorough history and physical examination can ascertain the diagnosis in the majority of patients who present with neurocardiogenic syncope, the yield of conventional diagnostic investigations is far lower in the case of syncope of cardiac origin. While a correlation between symptoms and electrocardiographic recordings is the gold standard, it remains difficult to confirm with traditional monitoring methods because of the sporadicity of the disease manifestations.
The ILR, a subcutaneous device implanted in the left subclavian region, enables the loop recording of a bipolar electrocardiographic channel for an extended ambulatory period. The device has a loop memory, which records and memorises the electrocardiogram (ECG) upon its activation by the patient or by a bystander following a symptomatic episode, as well as automatically on the basis of pre-programmed detection criteria. Several early studies have validated the performance of the implantable Holter for the evaluation of recurrent syncope that had remained unexplained despite extensive investigations. This device might also have a place earlier in the decision-making process for patients who present with more benign forms of syncope, with a view to avoid unnecessary and costly diagnostic tests. More recently, the diagnosis of atrial fibrillation (AF) was enabled by the addition of a discrimination algorithm similar to that used by implantable cardioverter defibrillators (ICD). The recordings triggered automatically or in the wake of an episode can be remotely transmitted by the CareLink telemedicine system, allowing a shortening of the time between diagnosis and onset of therapy. Over time, further progress might allow the recording of signals other than the ECG, such as the arterial pressure, enabling the monitoring of hemodynamic function. The miniaturization of implanted devices might also facilitate their implantation and broaden their indications.
This monograph describes 1) the technologic characteristics and the main current and future indications of this type of device, and 2) the broad principles of interrogation and programming, using the analysis of representative recordings from device recipients.
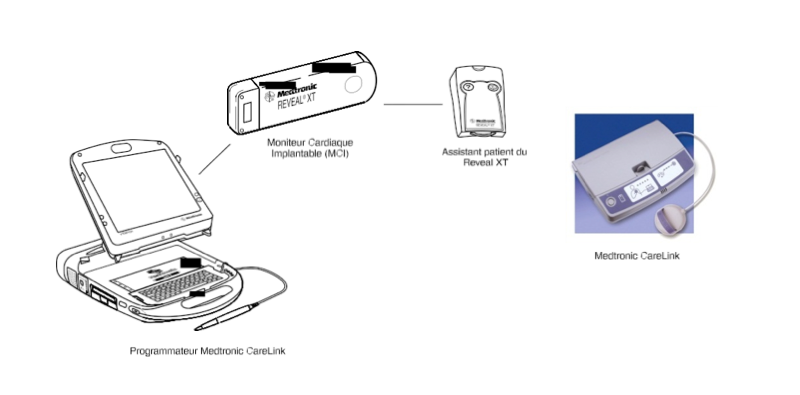
SYSTEM OVERVIEW
The system includes 4 elements that record the tracings and interrogate the device:
- The implantable heart monitor proper, a leadless device with 2 electrodes located under the can, which sense the ambulatory ECG continuously and on the long term. The most recent devices store in their memory 22.5 min of patient activated and 27 min of automatically sensed electrocardiographic recordings.
- The patient assistant, a portable, battery-powered telemetry device, which allows patients to memorise the tracings that coincide with a symptomatic episode.
- The Medtronic programmer, which allows programming of the device after its implantation and visualising the automatically recorded or patient-activated episodes.
- The CareLink telemedicine transmission system, which allows the remote transmission and speedy diagnosis of the episodes.
IMPLANTATION
Implantable Holters are usually implanted under local anaesthesia in the same operating suite as cardiac pacemakers. After wide disinfection of the left thoracic field, the device is placed between the first intercostal space and the 4th rib. The quality of the recordings, the prevention of oversensing noise that saturates the memories and the proper discrimination of the various types of arrhythmias hinge on an optimal implantation procedure.
Two steps must be given priority:
- The optimisation of R wave sensing. The kit includes the Reveal Vector Check, a mapping tool, which enables the selection of the optimal site without contaminating the device. The Vector Check has 2 electrodes approximately 4 cm apart, which corresponds to the space between the electrodes of the device. These electrodes are interfaced with the device at one end and are in contact with the skin of the patient at the other. The quality of the sensed signals can be verified, using the programmer. Minor variations in the device orientation may be associated with marked variations in amplitude and morphology of the sensed signals. A satisfactory position is associated with the sensing of a stable ≥0.3 mV R wave amplitude and morphology, satisfactory visualisation of P and R waves with high R/T and R/P ratios, and absence of P and T wave sensing by the device.
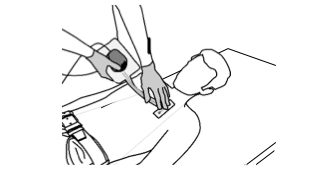
- Minimisation of the risk of oversensing myopotentials. To guarantee the reliability of automatic sensing of the episodes, motion of the device inside its pocket must be limited as much as possible. Therefore, the pocket must be neither too large, in order to limit movements, nor to small, in order to prevent the extrusion of the device. Suture holes in the pulse generator header can be used to attach the device to the subcutaenous tissues and minimise its rotation and migration after its implantation.
Once the device has been implanted, the sensing and data collection must be configured, and the patient assistant given and explained to the patient.
AUTOMATIC RECORDING OF THE EPISODES
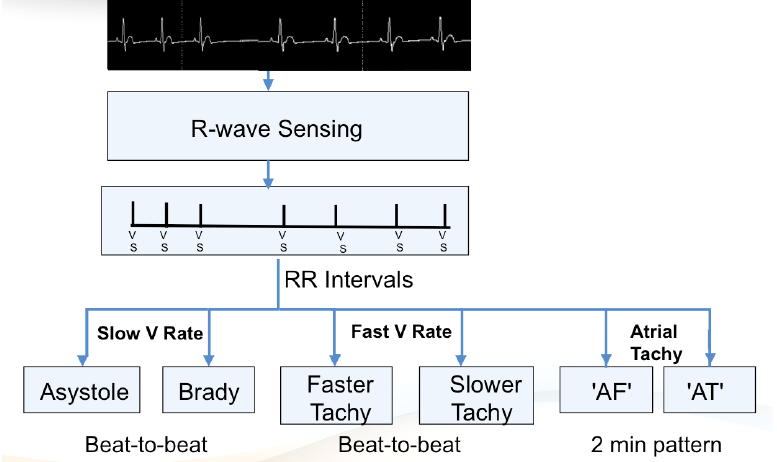
The last generation of ILR can classify and record 5 types of arrhythmic episodes:
- Ventricular tachycardia (VT): the heart rate increases above the programmed VT threshold.
- Fast ventricular tachycardia (FVT) or ventricular fibrillation (VF): the heart rate increases above the programmed FVT threshold.
- Bradyarrhythmia: the heart rate slows below the programmed bradyarrhythmic threshold.
- Asystole: no ventricular event is sensed during a programmed period.
- Atrial tachyarrhythmia (AT) or fibrillation (AF) or AF alone: The episodes of AF with atrioventricular conduction are diagnosed by analyzing the regularity of the ventricular rhythm, using an automatic algorithm.
Reveal XT, the most recent ILR, logs up to 30 arrhythmic episodes of each type. When the log is saturated, the data of the last episode replace those of the oldest of the same type.
The Reveal DX and XT can also store in the device memory the ECG recorded before and during the episode. For each episode of AT/AF, the Reveal XT stores the first 2 min of ECG. For each episode of VT, FVT, bradycardia or asystole, 30 sec of ECG before the onset, and up to 27 sec before the end of the episode are recorded.
The Reveal DX and XT store 27 min of ECG for the episodes sensed automatically. When the memory reserved for the episodes detected automatically is full, a new recording replaces the oldest stored ECG. The system replaces a recording only when ≥3 episodes of the same type remain in memory.
RECORDINGS OF EPISODES USING THE PATIENT ASSISTANT
Reveal DX and XT store up to 10 symptomatic, patient-activated episodes in the memory log. When the latter is full, the data of the latest episode replace those of the oldest. Reveal DX and XT reserve 22.5 min of ECG storage in the data log for the most recently recorded symptomatic episodes. The symptomatic episodes may be programmed to record
- 3 episodes, including 6.5 min before and 1 min after)
- 2 episodes, including 9 min before and 1 min after,
- a single episode including 14 min before and 1 min after activation of the system.
When the memory reserved for symptomatic episodes is full, the ECG of the oldest stored episode is replaced by the latest episode.
PROGRAMMING
Adjustment of the R wave sensing
The optimal placement of the ILR must allow the visualisation of the P, R and T waves to facilitate the interpretation of the recordings. On the other hand, the automatic sensing of bradycardia, VT as well as AF is strictly based on the count of the R wave and analysis of its regularity. The optimisation of the device position at the time of implantation is key, as all changes in its programming are unlikely to compensate for the recording of low quality signals. The programmable settings available to enable the sensing of R waves without sensing of either the P or T waves or the myopotentials include
- the sensitivity
- the refractory period and
- the duration of threshold stability before its decrease.
The sensing threshold defines the lowest electrical amplitude associated with sensing of an event. As in implantable cardioverter defibrillators (ICD), the sensing threshold, instead of being fixed, adapts automatically to the amplitude of the sensed R wave, and increases during the cardiac cycle. After sensing of an R wave, a programmable post-Vs refractory period is initiated and the sensing threshold is set at 65% of the R wave measured. This sensing threshold remains at this level for the entire programmable duration of threshold stability before decreasing. The sensing threshold keeps decreasing until a new R wave is sensed or the lowest threshold has been reached. The latter corresponds to the programmed setting of sensitivity.

The programming of the sensitivity must allow an accurate and flawless sensing of the R wave, including an adequate safety margin, while avoiding the sensing of P and T waves. The nominal value is 0.035 mV. The setting of the post Vs refractory period must prevent a double counting of the R wave. Programming of an excessively long refractory period limits the sensing of rapid rates by the device. The nominal value is 70 ms. The duration of threshold stability before its decrease must be optimised to avoid T wave sensing. The nominal value is 100 ms.
Configuration of automatic episodes sensing
FVT episodes
An event is classified as FVT when the ventricular cycle is shorter than the programmed FVT cycle (nominal = 300 ms). If the number of FVT events exceeds the programmed number (nominal = 12 / 16 FVT cycles), the device memorises an episode of FVT.
The FVT episode ends when one of the following criteria is fulfilled:
- Eight consecutive R waves are sensed with 1 cycle ≥ the programmed FVT cycle.
- The median ventricular cycle is ≥ the programmed FVT cycle during a 20 sec period.
- No R wave is sensed during a 10-sec period.
VT episodes
Un event is classified as VT when the ventricular cycle is shorter than the programmed VT cycle, though longer than the programmed FVT cycle (nominal value = 360 ms). If the number of VT events exceeds the programmed number (nominal = 16 consecutive cycles), an episode of VT is memorised.
The VT episode ends when one of the following criteria is fulfilled:
- Eight consecutive R waves are detected at a cycle ≥ the programmed VT cycle.
- The median ventricular cycle is ≥ the programmed VT cycle during a 20-sec period.
- No R wave is sensed during a 10-sec period.
To prevent the sensing of unstable ventricular rhythms during an AT/AF episode with a rapid ventricular response, or during sinus tachycardia, the sensing specificity may be increased by programming the stability and sudden onset settings.
• Stability: an R-R cycle capable of triggering the recording of an episode of VT is compared with 3 preceding VT cycles. If the difference between the current cycle and 1 of the 3 preceding cycles is > the programmed VT stability cycle, the R-R cycle is not classified as a VT event.
• Sudden onset: comparison between the 4 most recent and the 4 preceding RR cycles. If the mean duration of the 4 most recent RR cycles is < mean duration of the 4 preceding RR cycles, multiplied by the programmed percent sudden onset, the next 4 R-R cycles are classified as VT events.
Bradyarrhythmia episodes
An episode of bradyarrhythmia is diagnosed when the programmed number of RR cycles is > programmable duration. An RR cycle >2000 ms is nominally labelled as “Brady”.
An episode of bradyarrhythmia ends when one of the following criteria is fulfilled:
• Four consecutive R - R cycles are sensed, whose duration is ≤ programmed ventricular cycle;
• No R wave is sensed for ≥ 10 sec.
Episodes of asystole
An asystolic event begins when a cycle following a sensed R wave is longer than a programmed duration (nominal = 3 sec). Asystole ends after the sensing of 4 R waves.
Atrial tachyarrhythmias/atrial fibrillation
The episodes of AT/AF are diagnosed by means of an automatic algorithm based on the RR variability over a 2 min period. The differences among consecutive RR cycles are displayed on a Lorenz curve. The distribution of RR cycles allows the identification of the AT and AF episodes. During AF, the RR cycles are highly irregular and non-correlated in contrast with AT, when they are more regular.
Summary of arrhythmias detection:
INDICATIONS : BROAD PRINCIPLES
The ILR allows the ambulatory monitoring of the ECG for an extended period of time. A few important principles must be weighed when considering the implantation of this type of devices:
- A medical history and physical examination allow a determination of the origin of syncope in a majority of patients without further investigations.
- An initial risk stratification is essential to identify high-risk patients who have an indication for the implantation of a cardiac pacemaker or ICD.
- The ILR is indicated only for patients at high likelihood of suffering ≥1 symptomatic recurrence during the life of the device. A mean interval between symptoms is, therefore, a determining factor and should correspond to the characteristics of the device. The external event monitors are not useful in the investigation of syncope, as they are not able to record the ECG retrospectively. A patient who suffers weekly syncopal events should receive an external Holter for an extended period. Finally, the probability of diagnosis with an implantable Holter is low in a patient who has suffered a single episode of syncope or episodes separated by > 36 months, the battery life of the most recent devices.
- The gold standard is the recording of an ECG coinciding with the occurrence of symptoms, allowing the confirmation or exclusion of an arrhythmic cause. The automatic recording by the device of an episode of bradycardia or tachycardia in the wake of symptoms might yield useful information, though no firm diagnosis.
- An unequivocal diagnosis requires a recurrence, which may have disabling consequences, for example a facial trauma or a vehicular accident.
- Some channelopathies present with characteristics, which preclude the use of an implantable Holter. In Brugada syndrome, for example, the first arrhythmic episode is often life-threatening VF, for which the ILR, being only a witness, plays no therapeutic role.
IMPLANTABLE LOOP RECORDER AND SYNCOPE
When used as a diagnostic tool for unexplained syncope, the ILR allows the recording of an ECG in the wake of an episode.
DIAGNOSTIC YIELD
In an initial clinical study of the ILR in a highly selected patient sample who were at high risk of recurrent syncope and had suffered a mean of over 8 episodes, a correlation between symptoms and ECG was confirmed in 88% of patients over a 5-month period. The combined results of 9 studies, comprising over 500 patients presenting with unexplained syncope who had undergone extensive investigations, indicated a correlation of 35%, with asystole in 56%, tachycardia in 11%, and no arrhythmia in 33%. The yield depends on the patient selection and on the likelihood of recurrence, as well as on the life expectancy of the device’s batteries, which currently reach 36 months.
REPRODUCIBILITY OF THE EPISODES
The correlation between ECG and syncopal event is the gold standard as much as the diagnosis of the different episodes is the same. Both ISSUE studies showed that, in a majority of patients, the recorded ECG is highly reproducible among recurrences.
PATIENT GROUPS IN WHOM THE DIAGNOSTIC YIELD OF THE ILR IS HIGH
- Unexplained syncope in presence of a cardiac abnormality.
Various studies have found a value of the implantable Holter in the investigations of syncope in patients presenting with:
- bundle branch block and negative electrophysiologic studies;
- old myocardial infarction or cardiomyopathy with a depressed left ventricular (LV) ejection fraction (EF) or non-sustained VT and negative electrophysiologic studies;
- congenital heart disease.
- Identification of the mechanism of reflex syncope The merit of permanent pacing for malignant neurocardiogenic syncope remains controversial, though implies the demonstration of a cardioinhibitory mechanism and prolonged asystole. In contrast to the tilt-table test, the implantable Holter records the ECG in the wake of a spontaneous episode and allows an orientation of the therapy.
- Diagnosis of loss of consciousness under particular circumstances. The ILR allows a triage of the origin of repetitive loss of consciousness under particular circumstances, like recurrences of attacks labelled epileptic despite optimal treatment, episodes occurring in a context of major depressive state, or falls in elderly patients.
CLASSIFICATION OF SYNCOPE
The investigators of the ISSUE study proposed a classification of the recordings obtained by the ILR at the time of syncopal events.
- Type 1, corresponding to asystole, is observed in 63%,
- type 2, corresponding to bradycardia, in 5%,
- type 3, corresponding to absence of anomalous recording, in 18%, and
- type 4, corresponding to a tachycardia, in 14% of patients.
These 4 types can be subdivided, offering pathophysiologic information on the mechanism of syncope and allowing an orientation of the therapy. A reflex origin of syncope is suspected (types 1A, 1B and 2), in presence of progressive bradycardia, or a tachycardia followed by profound bradycardia, or in presence of asystole. In contrast, the sudden onset of complete atrioventricular block, without prior change in sinus rate or PR interval, suggests an intrinsic conduction disorder at the level of the His–Purkinje network (type 1C). In absence of arrhythmia at the time of syncope (type 3), a moderate slowing or acceleration of the sinus rate suggests an activation of the autonomic nervous system, a vasodilatory mechanism and a hypotensive syncopal event.
COST-EFFICTIVENESS OF A STRATEGY BASED ON THE ILR
The initial cost of the device is relatively high. However, the introduction of the implantable Holter early in the decision-making process allows the avoidance of often unnecessary and costly investigations, and lowers the cost / efficacy ratio.
WHERE TO PLACE THE ILR IN THE DECISION-MAKING PROCESS ?
At first, the ILR was used as a last recourse, after extensive investigations of recurrent syncope had remained negative. It now seems clear that the relatively low sensitivity and specificity of the conventional ambulatory ECG, tilt-table test, ATP test and electrophysiologic studies make them of low value in the investigations of syncope, and that the ILR might be worth considering as a first diagnostic choice. Various studies will show that the implantation of an ILR as a first choice is appropriate and offers a higher diagnostic and economical yield than conventional investigations, as long as high-risk patients are excluded.
An initial evaluation allows the identification of high-risk patients.
- In the majority of patients, the diagnostic is clear after a medical history and physical examination, and no further investigation is needed. The patients at low likelihood of suffering from syncope due to an arrhythmia are not candidates for the implantation of this type of device.
- Patients presenting with recurrent syncope and one of the following characteristics should be recommended urgent hospitalisation and detailed investigations: a) syncope on exertion, b) palpitation perceived at the time of syncope, c) family history of sudden death, d) non-sustained VT, bifascicular block or intraventricular conduction abnormalities in presence of wide QRS, e) inappropriate sinus bradycardia <50 bpm or sino-atrial block, f)ventricular preexcitation, short or prolonged QT interval, ECG changes consistent with Brugada syndrome or arrhythmogenic right ventricular dysplasia (negative T wave in the right precordial, epsilon wave and late ventricular potentials), or g) anaemia or abnormal blood electrolytes concentrations. The use of an ILR will be reserved for patients in whom all investigations have failed to yield a diagnosis.
- Patients presenting with a firm indication for an ICD should undergo the implant, using the device’s memory to identify the mechanism of syncope. Non-sustained VT in the context of a history of myocardial infarction is an indication for electrophysiologic studies and programmed ventricular stimulation.
- In these cases, as well as in patients whose ECG suggests the occurrence of paroxysmal bradycardia, who should undergo a period of ECG telemetry and, perhaps, electrophysiologic studies, the ILR should be used after all tests have remained negative.
- In absence of risk factors, however, the ILR can be used without prior investigations.
RECOMMENDATIONS
The 2009 European recommendations distinguish 2 class I and 1 class IIA indications for the implantable Holter as part of the investigations of syncope.
Class I
Early phase of evaluation in patients suffering from recurrent, unexplained syncope, in absence of risk factor and at high likelihood of sustaining a recurrence before depletion of the device’s battery.
Patients who have ≥1 risk factor and in whom detailed investigations have neither revealed the cause of syncope nor suggested a specific treatment.
Class IIA
The ILR might be considered for patients presenting with recurrent, malignant neurocardiogenic syncope to identify its mechanism and orient the therapy.
IMPLANTABLE ILR AND PALPITATION
Palpitation is a common symptom caused by a variety of cardiac rhythms, ranging from appropriate or inappropriate sinus tachycardia to life-threatening VT. The diagnosis is confirmed when the ECG can be recorded during an episode of palpitation, which, with conventional tests, may be challenging if the symptoms are few and far between. As for syncope, patients presenting with risk factors must be hospitalised and undergo extensive investigations. The benign health status found in the majority of patients complaining of palpitation limits the role played by the ILR. It might, therefore, be considered despite being invasive and costly only when a) the likelihood of making a diagnosis with conventional methods, is low, including ≥ 24-h of external, ambulatory ECG, b) the symptoms are major and consistent with circulatory dysfunction, and c) all investigations have remained negative.
RECOMMENDATIONS
Class IIA
The ILR can be considered for carefully selected patients presenting with infrequent palpitation associated with major symptoms, after an external, ambulatory ECG of 24 h or longer has not yielded a diagnosis.
ILR AND ATRIAL FIBRILLATION
The diagnosis of AF and the evaluation of the efficacy of its therapy might represent an important upcoming area of investigation for the ILR. If a heart rate control strategy has been chosen, no complicated investigation is necessary, as the patient follow-up is based on the clinical status. On the other hand, if a rhythm control strategy has been chosen, a precise knowledge of the occurrence, duration and frequency of AF recurrences is essential to adjust the antiarrhythmic and antithrombotic treatments. An accurate evaluation of the duration of the episodes of paroxysmal AF seems also critical, as episodes lasting > 24 h are associated with an increased risk of thromboembolic complications. Several studies have found a weak correlation between symptoms and ECG confirmation of AF. Indeed, symptoms were perceived in only 46% of episodes of AF confirmed electrocardiographically. Recurrences of AF may be asymptomatic in patients previously symptomatic. Furthermore, sinus tachycardia or repetitive extrasystoles might be erroneously perceived as recurrences, causing an overestimation of the AF burden. The correlation is particularly weak when a rhythm control strategy has been chosen. The memory of early devices, which were not equipped with a specific algorithm, was saturated by the sensing of myopotentials, precluding the automatic analysis of episodes of atrial arrhythmias and an accurate quantification of the arrhythmic burden. Technologic progress was therefore needed to reliably differentiate sinus rhythm from atrial arrhythmias. The newest devices include an algorithm similar to that used in ICD, which identify episodes of atrial arrhythmias on the basis of rhythm irregularity. Several large studies are examining the performance of this type of device in the qualitative and quantitative diagnosis of AF episodes as well as in their ability to identify the source of cryptogenic cerebral vascular accidents.
RECOMMENDATIONS
No guideline has been issued by the European Society of Cardiology with regard to the monitoring or diagnosis of AF with the ILR.
PERSPECTIVES
Several technologic advances and the completion of studies including new patient profiles should allow the broadening of the indications and implantation of this type of device.
Further miniaturisation of the device will facilitate its implantation and acceptance by patients and caregivers. Despite the indications clearly formulated by professional societies, the ILR is currently largely underutilised. The invasive nature of the implant procedure, which must be performed in an electrophysiology laboratory may, at least partially explain the discrepancy between indications and actual device implantations. The very small size of upcoming devices should allow their implant in an ambulatory setting, in a non-specialized environment, and should minimize their aesthetic consequences.
The device’s life expectancy is directly correlated with the probability of recording a spontaneous symptomatic episode. The 36-month life expectancy of current batteries might increase considerably in the near future.
The information collected by the device can be remotely transmitted by the CareLink system. Its adaptation by the medical centres to the follow-up of ILR should shorten the delays between diagnosis and introduction of therapy, when indicated. The remote follow-up and ambulatory management of AF, and the decision of introducing or discontinuing antithrombotic therapy might represent an important application of the system.
New hemodynamic sensors could be added to the functions of the ILR, allowing the follow-up of heart failure and adaptation of its treatment. Furthermore, the monitoring of arterial pressure might contribute key information toward the identification of the mechanism(s) of syncope and allow the continuous surveillance of the efficacy of treatment of arterial hypertension.
Progress in the morphologic analysis of ECG signals should allow the follow-up of changes in ST segment and the detection of episodes of silent ischemia.
Randomized studies are needed to determine the optimal place of the ILR in the diagnosis of unexplained syncope and identification of the causes of palpitation.



